Palladium
Background to the schools Wikipedia
Arranging a Wikipedia selection for schools in the developing world without internet was an initiative by SOS Children. Click here to find out about child sponsorship.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General data | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Name, symbol, number | palladium, Pd, 46 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Element category | transition metals | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group, Period, Block | 10, 5, d | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | silvery white metallic |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard atomic weight | 106.42 (1) g·mol−1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Kr] 4d10 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 18, 18, 0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase | solid | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density (near r.t.) | 12.023 g·cm−3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Liquid density at m.p. | 10.38 g·cm−3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | 1828.05 K (1554.9 °C, 2830.82 °F) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | 3236 K (2963 °C, 5365 °F) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of fusion | 16.74 kJ·mol−1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Heat of vaporization 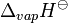 |
362 kJ·mol−1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Specific heat capacity | (25 °C) 25.98 J·mol−1·K−1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal structure | cubic face centered | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxidation states | 2, 4 (mildly basic oxide) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electronegativity | 2.20 (Pauling scale) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ionization energies | 1st: 804.4 kJ/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2nd: 1870 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3rd: 3177 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic radius | 140 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic radius (calc.) | 169 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Covalent radius | 131 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Van der Waals radius | 163 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Miscellanea | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Magnetic ordering | no data | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrical resistivity | (20 °C) 105.4 n Ω·m | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal conductivity | (300 K) 71.8 W·m−1·K−1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal expansion | (25 °C) 11.8 µm·m−1·K−1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Speed of sound (thin rod) | (20 °C) 3070 m/s | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Young's modulus | 121 GPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shear modulus | 44 GPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bulk modulus | 180 GPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Poisson ratio | 0.39 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mohs hardness | 4.75 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vickers hardness | 461 MPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Brinell hardness | 37.3 MPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS registry number | 7440-05-3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Most-stable isotopes | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Palladium (pronounced \pe-‘lä-dē-em\) is a rare and lustrous silvery-white metal that was discovered in 1803 by William Hyde Wollaston, who named it palladium after the asteroid Pallas, which in turn, was named for the Greek Goddess of Wisdom. The symbol for palladium is Pd, and its atomic number is 46. Palladium, along with platinum, rhodium, ruthenium, iridium and osmium form a group of elements referred to as the platinum group metals (PGMs). PGMs share similar chemical properties, but palladium is unique in that it has the lowest melting point and is the least dense of these precious metals. Incredibly, when palladium is at room temperature and atmospheric pressure, it can absorb up to 900 times its own volume of hydrogen, which make palladium an efficient and safe storage medium for hydrogen and hydrogen isotopes. Palladium is also tarnish resistant, electrically stable and resistant to chemical erosion as well as intense heat.
The unique properties of palladium and other PGMs necessitate their widespread use as one in four of the goods manufactured today either contain PGMs or had PGMs play a key role during the manufacturing process. Over half of the supply of palladium and its sister metal platinum goes into catalytic converters, which convert up to 90% of harmful gases from auto exhausts ( hydrocarbons, carbon monoxide and nitrogen oxide) into less harmful substances (nitrogen, carbon dioxide and water vapor). Palladium’s precious metal qualities and appearance generate significant consumption in the luxury jewelry market. Palladium is found in many electronics including computers, mobile phones, multi-layer ceramic capacitors, component plating, low voltage electrical contacts, and SED/OLED/LCD televisions. Palladium is also used in dentistry, medicine, hydrogen purification, chemical applications, groundwater treatment, and it is playing a key role in the technology used for fuel cells, which combine hydrogen and oxygen to produce electricity, heat and water with virtually no pollution.
Ore deposits of palladium and other PGMs are rare, and the most extensive deposits have been found in the norite belt of the Bushveld Igneous Complex in the Transvaal in South Africa, the Stillwater Complex in Montana, USA, the Sudbury District of Ontario Canada and the Norilsk Complex in Russia. Besides mining, palladium is also recovered through recycling; the bulk of which comes from scrapped catalytic converters. The numerous applications and limited supply sources of palladium result in palladium drawing considerable investment interest.
History
Palladium was discovered by William Hyde Wollaston in 1803. This element was named by Wollaston in 1804 after the asteroid Pallas, which was discovered two years earlier.
Wollaston found palladium in crude platinum ore from South America by dissolving the ore in aqua regia, neutralizing the solution with sodium hydroxide, and precipitating platinum as ammonium chloroplatinate with ammonium chloride. He added mercuric cyanide to form the compound palladium cyanide, which was heated to extract palladium metal.
Palladium chloride was at one time prescribed as a tuberculosis treatment at the rate of 0.065g per day (approximately one milligram per kilogram of body weight). This treatment did have many negative side-effects, and was later replaced by more effective drugs.
Palladium's affinity for hydrogen led it to play an essential role in the Fleischmann-Pons experiment in 1989, also known as cold fusion.
In the run up to 2000, Russian supply of palladium to global market was repeatedly delayed and disrupted because the export quota was not granted on time, due to political reasons. The ensuing market panic buying drove the palladium price to an all time high of $1100 per ounce, reached in January, 2001. During the time period, Ford Motor Company, in fear of auto vehicle production disruption due to a possible palladium shortage, stockpiled large amounts of the metal, purchased near the price high. As prices subsequently fell in early 2001, Ford lost nearly $1 billion U.S. dollars.
World demand for palladium increased from 100 tons in 1990 to nearly 300 tons in 2000. The global production from mines was 222 metric tons in 2006 according to USGS data. Most palladium is used for catalytic converters in the automobile industry.
Occurrence
In 2005, Russia was the top producer of palladium with at least 50% world share followed by South Africa, USA and Canada, reports the British Geological Survey.
Palladium is found as a free metal and alloyed with platinum and gold with platinum group metals in placer deposits of the Ural Mountains, Australia, Ethiopia, South and North America. It is commercially produced from nickel-copper deposits found in South Africa, Ontario, and Siberia; the huge volume of ore processed makes this extraction profitable despite the low proportion of palladium in these ores. The world's largest single producer of palladium is MMC Norilsk Nickel produced from the Norilsk–Talnakh nickel deposits. The Merensky Reef of the Bushveld Igneous Complex of South Africa contains significant palladium in addition to other platinum group elements. The Stillwater igneous complex of Montana also contains mineable palladium.
Palladium is also produced in nuclear fission reactors and can be extracted from spent nuclear fuel, see Synthesis of noble metals, though the quantity produced is insignificant.
Palladium is found in the rare minerals cooperite and polarite.
Characteristics
Palladium is a soft silver-white metal that resembles platinum. It is the least dense and has the lowest melting point of the platinum group metals. It is soft and ductile when annealed and greatly increases its strength and hardness when it is cold-worked. Palladium is chemically attacked by sulfuric, nitric, and hydrochloric acid in which it dissolves slowly. This metal also does not react with oxygen at normal temperatures (and thus does not tarnish in air). Palladium heated to 800°C will produce a layer of palladium(II) oxide (PdO). It lightly tarnishes in moist atmosphere containing sulfur.
This metal has the uncommon ability to absorb up to 900 times its own volume of hydrogen at room temperatures. It is thought that this possibly forms palladium hydride (PdH2) but it is not yet clear if this is a true chemical compound.
When palladium has absorbed large amounts of hydrogen, it will expand slightly in size.
Common oxidation states of palladium are 0,+1, +2 and +4. Although originally +3 was thought of as one of the fundamental oxidation states of palladium, there is no evidence for palladium occurring in the +3 oxidation state; this has been investigated via X-ray diffraction for a number of compounds, indicating a dimer of palladium(II) and palladium(IV) instead. Recently, compounds with an oxidation state of +6 were synthesised.
Isotopes
Naturally-occurring palladium is composed of six isotopes. The most stable radioisotopes are 107Pd with a half-life of 6.5 million years, 103Pd with a half-life of 17 days, and 100Pd with a half-life of 3.63 days. Eighteen other radioisotopes have been characterized with atomic weights ranging from 92.936 u (93Pd) to 119.924 u (120Pd). Most of these have half-lives that are less than a half-hour, except 101Pd (half-life: 8.47 hours), 109Pd (half-life: 13.7 hours), and 112Pd (half-life: 21 hours).
The primary decay mode before the most abundant stable isotope, 106Pd, is electron capture and the primary mode after is beta decay. The primary decay product before 106Pd is rhodium and the primary product after is silver.
Radiogenic 107Ag is a decay product of 107Pd and was first discovered in the Santa Clara, California meteorite of 1978. The discoverers suggest that the coalescence and differentiation of iron-cored small planets may have occurred 10 million years after a nucleosynthetic event. 107Pd versus Ag correlations observed in bodies, which have clearly been melted since accretion of the solar system, must reflect the presence of short-lived nuclides in the early solar system.
Applications
Palladium is used in dentistry, watch making, in blood sugar test strips, in aircraft spark plugs and in the production of surgical instruments and electrical contacts. Palladium is also used to make professional transverse flutes.
Electronics
The biggest application of palladium in electronics is making the multilayer ceramic capacitor. Palladium (and palladium-silver alloys) are used as electrodes in multi-layer ceramic capacitors. Palladium (sometimes alloyed with nickel) is used in connector platings in consumer electronics.
It is also used in plating of electronic components and in soldering materials. The electronic sector consumed 1.07 million troy ounces of palladium in 2006, according to a Johnson Matthey report.
Technology
Hydrogen easily diffuses through heated palladium; thus, it provides a means of purifying the gas. Membrane reactors with Pd membranes are therefore used for the production of hydrogen.
It is as part of the palladium-hydrogen electrode in electrochemical studies. Palladium (II) chloride can absorb large amounts of carbon monoxide gas, and is used in carbon monoxide detectors.
Catalysis
When it is finely divided, such as in palladium on carbon, palladium forms a good catalyst and is used to speed up hydrogenation and dehydrogenation reactions, as well as in petroleum cracking. A large number of carbon-carbon bond forming reactions in organic chemistry (such as the Heck and Suzuki coupling) are facilitated by catalysis with palladium compounds. The largest use of palladium today is in catalytic converters.
Pd is also a versatile metal for homogeneous catalysis. It is used in combination with a broad variety of ligands for highly selective chemical transformations.
Hydrogen storage
Palladium hydride is metallic palladium that contains a substantial quantity of hydrogen within its crystal lattice. At room temperature and atmospheric pressure, palladium can absorb up to 935 times its own volume of hydrogen in a reversible process. This property has been investigated because hydrogen storage is of such interest and a better understanding of what happens at the molecular level could give clues to designing improved metal hydrides. A palladium based store, however, would be prohibitively expensive due to the cost of the metal.
Jewelry
Palladium itself has occasionally been used as a precious metal in jewelry, as replacement for platinum or white gold. This is due to its naturally white properties giving it no need for a rhodium plating. It is slightly whiter, much lighter and about 12% harder. Similar to gold, palladium can be beaten into a thin leaf form as thin as 100 nm (1/250,000 in). Like platinum, it will develop a hazy patina over time. Unlike platinum, however, palladium will discolor at soldering temperatures, become brittle with repeated heating and cooling, and react with strong acids.
It can also be used as a substitute for nickel when making white gold. Palladium is one of three most-used metals that can be alloyed with gold to produce white gold. (Nickel and silver can also be used.) Palladium-gold is a much more expensive alloy than nickel-gold but is hypoallergenic and holds its white colour better.
When platinum was declared a strategic government resource during World War II, many jewelry bands were made out of palladium. As recently as September 2001, palladium was more expensive than platinum and rarely used in jewelry also due to the technical obstacle of casting. However the casting problem has been resolved, and its use in jewelry has increased because of a large spike in the price of platinum and a drop in the price of palladium.
Prior to 2004, the principal use of palladium in jewelry was as an alloy in the manufacture of white gold jewelry, but, beginning early in 2004 when gold and platinum prices began to rise steeply, Chinese jewelers began fabricating significant volumes of palladium jewelry. Johnson Matthey estimated that, in 2004 with the introduction of palladium jewelry in China, demand for palladium for jewelry fabrication was 920,000 ounces, or approximately 14% of the total palladium demand for 2004, an increase of almost 700,000 ounces from 2003. This growth continued during 2005, with estimated worldwide jewelry demand for palladium of about 1.4 million ounces, or almost 21% of net palladium supply, again with most of the demand centered in China.
Photography
With the platinotype printing process photographers make fine-art black-and-white prints using platinum or palladium salts. Often used with platinum, palladium provides an alternative to silver.
Art
Palladium leaf is one of several alternatives to silver leaf used in manuscript illumination. The use of silver leaf is problematic due to its predisposition to tarnish. Aluminum leaf is a very inexpensive alternative, however aluminium is much more difficult to work than gold or silver and results in less than optimal results when employing traditional metal leafing techniques, and so palladium leaf is considered the best substitute despite its considerable cost. Platinum leaf may be used to the same effect as palladium leaf with similar working properties, but it is not as readily available in leaf form commercially.